Colloïdes et Hybrides
Silica-based delivery systems for oral delivery of drugs, enzymes and probiotics |
According to the US FDA and the European FSA, amorphous forms of silica and silicates are generally recognized to be safe as oral delivery ingredients in amounts up to 1.5 g per day. Silica is used in the formulation of solid dosage forms, e.g. tablets, as glidant or lubricant.
 The main challenge is to maintain the activity of the upload (particularly sensitive in the case of enzymes or bacteria) during encapsulation, processing, storage and even digestion (or passage through the gastrointestinal tract).
The main challenge is to maintain the activity of the upload (particularly sensitive in the case of enzymes or bacteria) during encapsulation, processing, storage and even digestion (or passage through the gastrointestinal tract).
Encapsulation involving sol-gel processes appears as a promising method to elaborate silica carriers for oral delivery application. [1] This is due silica’s biocompatibility, mechanical stability and ease of synthesis, at temperatures even below 40 °C, which makes it compatible with biomolecules or cells. We are interested in using colloidal systems (micelles and solid lipid nanoparticles) to both nanostructure the silica matrix and to encapsulate bioactive ingredients in order to obtain hybrid carriers for oral delivery applications. The encapsulation of three systems is highlighted: (a) a drug, curcumin [2], (b) an enzyme, b-Galactosidase [3] and (c) a probiotic, L. rhamnosus GG [4].
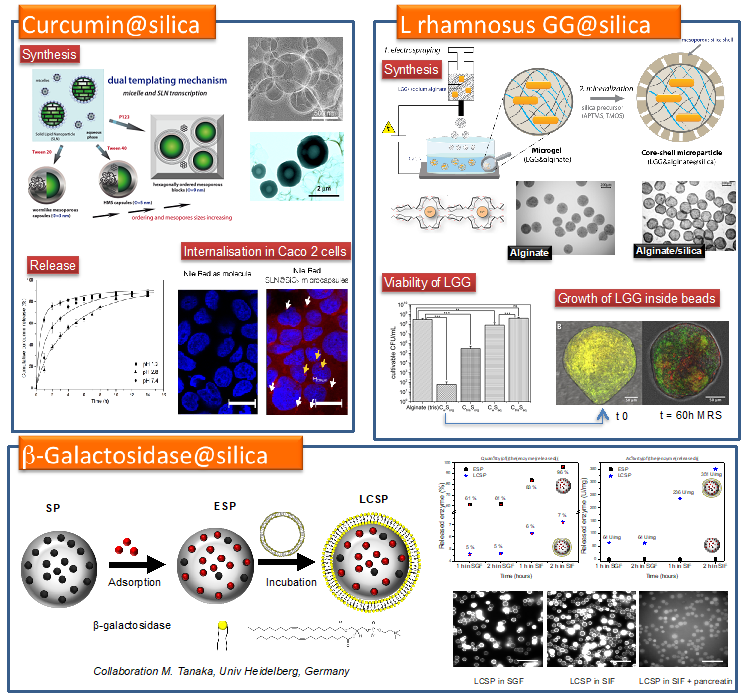 |  |
[1] R. Diab, N. Canilho, I. A. Pavel, F. B. Haffner, M. Girardon, A. Pasc Adv. Colloid Interface Sci. (SI : Biocolloidnanotechnology) 2017, 249, 346-362
[2] (a) Kim, S., Diab, R., Joubert, O., Canilho, N., Pasc, A. Colloids Surf. B Biointerfaces, 2016, 140, 161, (b) Kim, S., Stébé, M. - J., Blin, J. - L., Pasc, A., J. Mater. Chem. B, 2014, 2, 7910.
[3] (a) I.-A. Pavel, S. F. Prazeres, G. Montalvo, C. Garcia Ruiz, V. Nicolas, A. Celzard, F. Dehez, L. Canabady-Rochelle, N Canilho, A. Pasc Langmuir 2017, 33, 13, 3333, (b) I.-A. Pavel, M. Girardon, S. El Hajj, S. Parant, F. Amadei, S. Kaufmann, M. Tanaka, V. Fierro, A. Celzard, N. Canilho, A. Pasc J. Mater. Chem. B, 2018, 6, 5633.
[4] (a) Haffner, F. B., Girardon, M., Etienne, M., Fontanay, S., Canilho, N., Duval, R. E., Mierzwa, M., Diab, R., Pasc, A. J. Mater. Chem. B, 2016, 4, 7929, (b) Haffner, F. B., van de Wiele, T., Pasc A. J. Mater. Chem. B, 2017, 5, 7839, (c) F. B. Haffner, Pasc A., LWT - Food Sci. Technol. 2018, 91, 175.
retour vers accueil MolSyBio

